According to the statistics from the National Bureau of Statistics, the scale of Chinese health care products market is rising steadily. It is a great opportunity for oversea health food companies to enter the Chinese market because of the huge market demand and government’s favorable policies. Before putting products into the Chinese market, knowing the development status of health food and having an in-depth analysis of approved health food will be very helpful for product positioning and market entry time choosing for oversea health food companies.
China has gradually established a standardized management system and market for health food after about 30 years’ developing. Since the "Administrative Measures for Health Food" was implemented on June 1, 1996, the legal status of health food was recognized for the first time in the law, and a unified approval standard was established. There have been 17,279 approved health foods by the end of 2017, respectively, 16,494 approved domestic health foods and 785 approved imported health foods. The details are as follows:
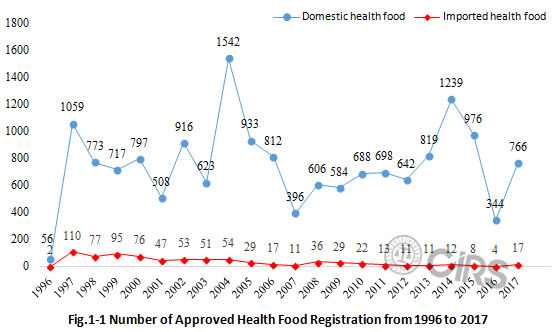
As
shown in Fig.1-1, although the amount of approved health foods is very large, the
annual approval number is fluctuating, with a maximum of 1542 and minimum of
56. According to
analysis by CIRS, policy change is the key reason for the significant increase and
decrease in the approval number and registration duration of health food. The
release and implementation of the new policy will affect the health food
approval progress of competent authority. Whereas, the
registration duration will have direct impact on the progress of health food
launch. Section 2 of
In addition, in order to assist oversea health food companies in understanding the development status of health food registration and choosing the product positioning more accurately, the data and analysis of approved health food from different angles will be presented in Section 2 of the Guideline. For example,
- Angle 1: Functional health food VS Vitamin and mineral nutrition supplement;
- Angle 2: 27 health functions for functional health food;
- Angle 3: Top 10 domestic enterprises and oversea manufacturers in obtaining health food registration certificates.
Finally, since the implementation of health food filing in July 2017, the number of approved health food filing was varying in dosage forms, nutrients, etc. Section 2 of the Guideline will make statistical analysis of health food filing data, which will help companies better understand the categories and distribution of approved health food filling.
The summary of each section in the Guideline is introduced by a series of articles which are available as following. If it is of your interest, please kindly click to get information.
Section 1: Information to Know before Health Food Exportation to China
Section 2: How Many Health Food are Registered in China?
Section 3: Chinese Health Food Regulation in Present and Future
Section 4: Timeline and Budget for Health Food Access to Chinese Market
Section 5: How does Chinese Government Supervise the Health Food?
In addition,
if you would like to get the
Editor:
CIRS Food Technical Team: Established in 2012, CIRS Food Technical Team has more than 80% masters with degrees of Food Safety or Food Engineering and more than 50% members from oversea renowned universities. Since it was founded, based on the rich experience of regulatory compliance and deep understanding of Chinese food regulations and industry, food specialists have made comprehensive and reasonable solutions for many oversea food companies to complete Chinese food regulatory compliance progress.
If you have any other questions, please contact us at service@cirs-group.com.
Related Article:
Cross-Border Acquisition: Chinese-funded Enterprises Frequently Acquire Foreign Health Food Brand
Data Analysis: Why are Health Food Enterprises Keen to Explore E-Commerce Channel?

