Since 2013, the amount of imported food in China has been increasing dramatically. According to China customs, there are many imported food products which are not meeting the requirements of Chinese regulations. Over 60% of the unqualified imported food products are general food products. The General Administration of Customs China (GACC) reserves the right to destroy or reject all products which are not fully compliant with Chinese regulations. Therefore, it is necessary to know the regulations your product needs to comply with before exporting to China.
Pre-packaged general food refers to foods which is prepackaged or made up with a measured quantity in a container. They should have unified labeling of quality or volume within a certain range.
When preparing your products, CIRS
technical team suggests that enterprises can follow the below 5 steps:
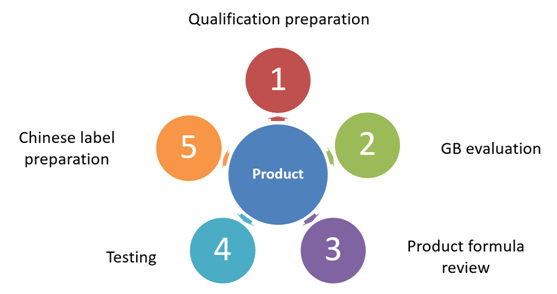
Qualification preparation:
Information filing for both exporter and importer,
Overseas manufacturer registration at GACC,
Other specific certifications for certain foods.
GB evaluation:
The purpose of this step is to conform with the GB standards of the products, ingredients, and production processes.
Product formula review:
CIRS suggests reviewing the product formula to ensure that each ingredient is allowed to be used and the quantities are within the limitation in the relevant GB standard.
Testing:
GB (China National Standard) tests includes sensory tests, physicochemical tests, microbiological etc.
A Nutrition test includes tests related to energy, protein, fat, carbohydrate, sodium, and other nutrients if applicable.
Please find the following common questions relating to General Food Products compliance in China for your reference:
1. What are the mandatory contents of nutrition labels for imported pre-packaged food?
The nutrition labels on imported pre-packaged food in China are usually presented in the form of a nutrition facts sheet. The mandatory labeling includes content and its percentage to the nutrient reference value of the energy, protein, fat, carbohydrate, and sodium.
(How to Make Compliant Nutrition Information for Prepackaged Common Food?)
2. Is ISO deemed equivalent to GB for product release into China?
No, ISO does not equal to GB in China. They are different regulations. All tests need to follow Chinese regulations and requirements for exporting products to China.
3. Do I need to conduct the nutrition test in a specific lab in China? Or can I do it oversea? and is the GB test only conducted for the initial import?
For the nutrition test, there is no specific lab requirement. The tests need to follow Chinese regulatory requirements. It may be done overseas, but we suggest all tests are carried out in a lab in China. This is because the report is in the Chinese language and therefore simplifies the custom check procedure in China. For most food categories, the GB test report is not mandatory to provide when importing. For some special food categories such as dairy products and vegetable oil, the GB test report is mandatory to provide. CIRS suggests conducting the GB test before importing to ensure that the product quality can comply with the requirements in China.
In line with inquiries we have received and experiences of our technical team, we have created a checklist to aid companies in complying with the regulations for exporting pre-packaged general food to China.
- Confirm the applicable GB (China National Standard) of the product (Prepackaged Food Regulations)
- Check ingredients are permitted and that quantities are within prescribed limits under the relevant GB standard (Formula Review on Prepackaged Food)
- Carry out nutrition tests and GB tests to ensure the product’s quality
- Review and revise the original label, translate the original label into Chinese (Common Errors in Chinese Labels of Prepackaged General Foods)
- Prepare a standard Chinese label under the China National Standard (Interpretation of Major Changes in GB 7718 Draft)
- Apply the information filing on Overseas manufacturers, Overseas Exporters/Agents, and Importers with GACC
- Register the overseas food manufacturers site with GACC (for dairy products, seafood products, meat products, and bird’s nest products)
If you have any further questions or inquiries, please contact CIRS Europe:
Address: Regus Harcourt Centre, Dublin 2, Ireland, D02 HW77
Tel: 00 353 (1) 477 3709
Email: service@cirs-group.com
Author

Ran Liu, Food Regulatory Consultant, CIRS Europe
Ms. Ran Liu graduated from University College Dublin with a MEngSc degree, majoring in Food Engineering. She has an extensive background in food ingredients, regulations, and process engineering. After joining the CIRS Group, she focuses on the food regulations, including general food, health food, and Foods for Special Medical Purpose (FSMP). She is currently working for CIRS Europe and responsible for providing food regulatory technical support for the European food and food-related companies.
Related Information
Free Webinar: How to Export Pre-packaged General Food to China

